I visited my grandmother in Greece this summer. Along with bags of food and candy, she handed me soap bars she had made. The smell pulled me back to childhood, when I showered after football with the same olive oil soap. She still uses nothing else.
This time, I asked her how she makes it. She listed three ingredients: extra virgin olive oil, water, and lye (sodium hydroxide). Simple. Almost too simple. None of those things can clean on their own. Yet together, they become soap.
That struck me. We all use soap every day. It covers our sinks and showers. It’s in every supermarket. But how does it actually work? And why are there so many kinds—bars, liquids, hand soaps, shampoos, dish soaps?
How Does Soap Clean?
When you mix olive oil, water, and potassium hydroxide, a remarkable reaction occurs. Through a reaction called saponification, fats and lye transform into soap. Three simple ingredients become a new substance with powers none of them had on their own.
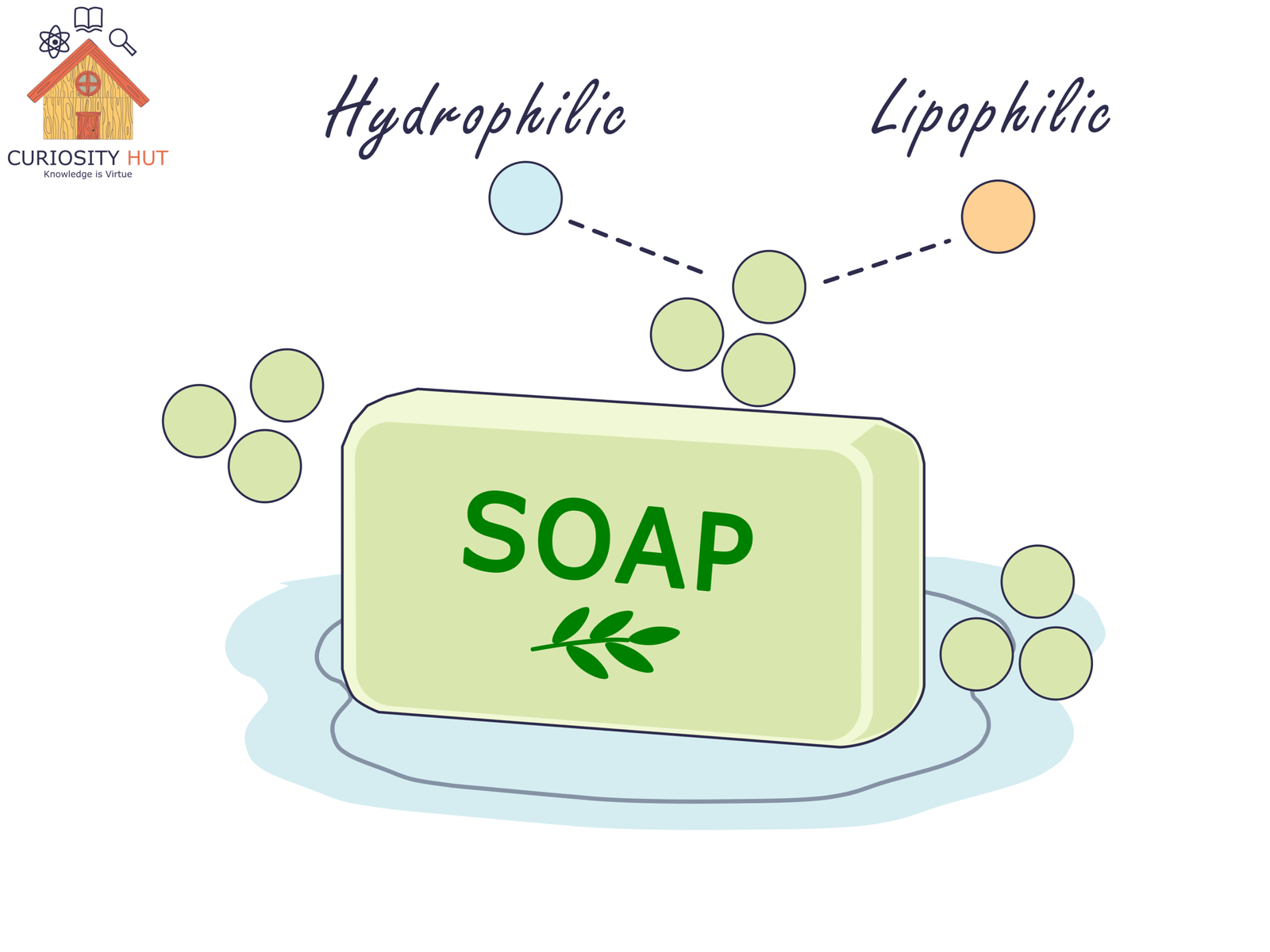
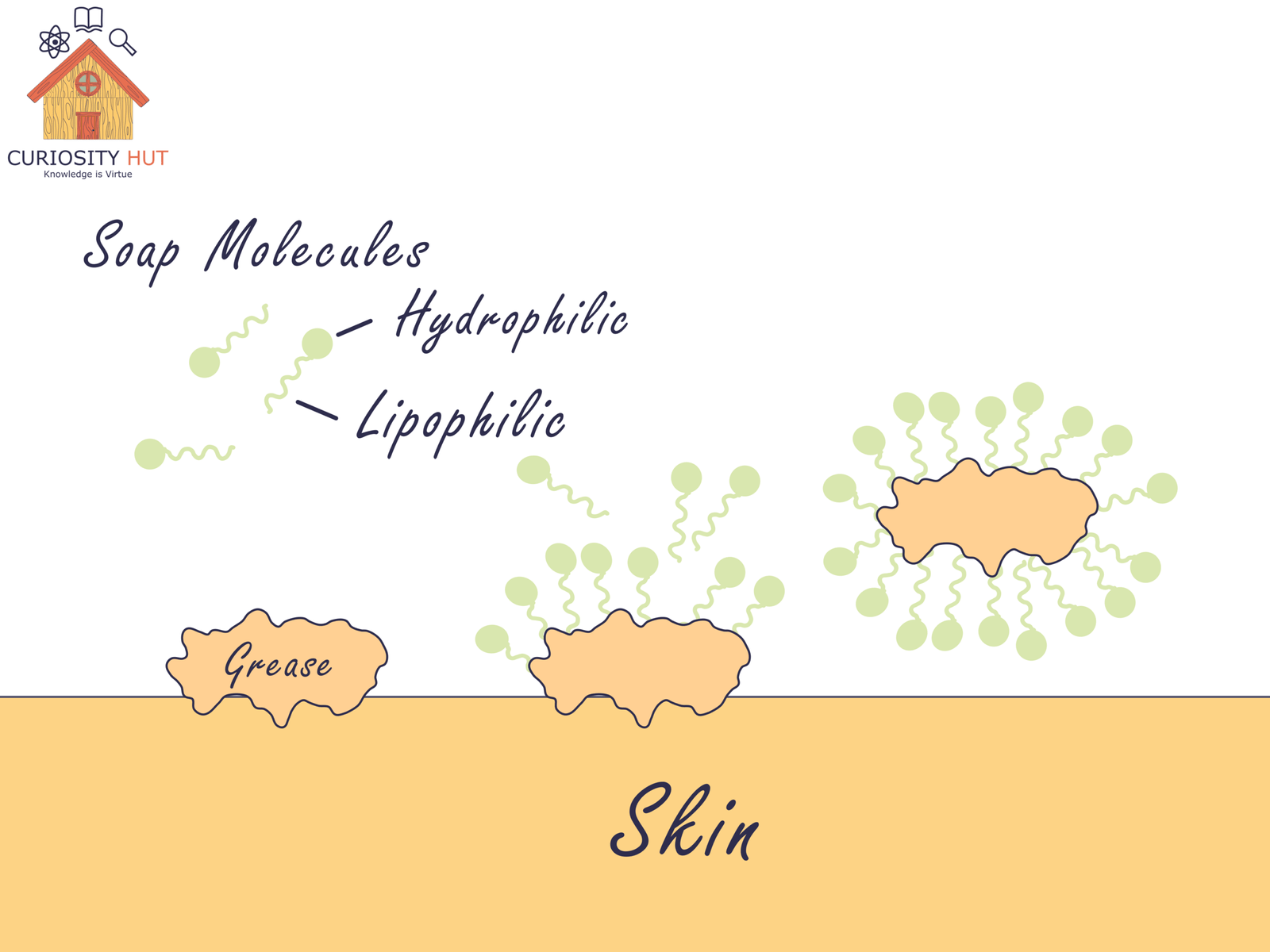
Soap molecules are double-sided. One end is lipophilic—it loves oils, grease, and the fatty coats of bacteria and viruses. The other end is hydrophilic—it loves water. This dual nature lets soap break through the standoff between oil and water. It binds to grime with one hand and pulls it away with the other, so water can wash it off your skin.
Soap also lowers water’s surface tension. That makes water spread out and slip into cracks and pores, carrying dirt away. Without soap, water beads up and leaves grease untouched.
What about antibacterial soap? Most of those products aren’t much better than plain soap. The “antibacterial” chemicals need time on your skin to work. Unless you keep lathering for many seconds, they won’t add much. Regular soap is effective at removing bacteria by scrubbing them off your hands. If you can’t wash at all, that’s when hand sanitizer earns its place.
Subscribe to my Free Newsletter
Sign up for blog updates, science stories, riddles, and occasional musings. No algorithms—just me, writing to you.
Not All Soaps Are the Same
Your grandmother’s bar is as old-school as it gets: olive oil mixed with lye and left to cure. Bar soaps usually use sodium hydroxide, which makes them solid. Liquid soaps swap in potassium hydroxide, producing a softer paste. From there, companies add fragrances, colors, or moisturizers. The core chemistry stays the same, but the texture and extras change.
Specialized soaps go further. Dish soap is designed to strip oils fast, even the stubborn film left on a frying pan. It’s tough on grease, but harsh on skin if you use it too often. Shampoo takes the opposite approach. It uses milder surfactants—soap-like molecules. They clean without stripping away all the natural oils your scalp needs. Conditioners and proteins are often mixed in to protect hair. Hand soaps sit somewhere in the middle. Strong enough to remove dirt but usually blended with softeners like glycerin so they don’t dry out skin.
Different recipes, same principle: molecules that grab onto oils and let water wash them away. The balance depends on what you’re trying to clean—your hands, your hair, or last night’s lasagna pan.
Soap and Your Skin
Soap does its job by binding to oils and washing them away. But your skin needs some of those oils. Strip too much, and your hands feel tight and dry. That’s why soaps often add glycerin or natural oils—they help lock in moisture and soften the effect.
Olive oil soaps, like the ones your grandmother makes, are rich in glycerin by nature. They cleanse without leaving skin raw. Modern “moisturizing soaps” use the same idea: balance cleaning power with ingredients that protect the skin barrier.
This balance matters. Too harsh, and soap damages the very surface it’s meant to protect. Too mild, and it won’t clean well enough. The sweet spot is a bar or liquid that removes grime but leaves enough natural oils behind to keep skin healthy.
Conclusion – More Than Just Soap
That bar of olive oil soap from my grandmother wasn’t just a gift. It was a piece of chemistry, culture, and memory. Three simple ingredients—oil, water, and lye—transform into something that breaks the rules of oil and water, washes away dirt, and even strips viruses of their coats.
We reach for soap every day without thinking. Yet inside every bar or bottle is a molecule with a double life: one side clings to grease and germs, the other drags them into water. Whether it’s shampoo, dish soap, or a rough green bar from a village in Greece, the principle stays the same.
The next time you lather up, remember you’re holding more than a cleaning product. You’re holding the quiet power of chemistry—something that keeps us alive, healthy, and connected to traditions older than history.
Subscribe to my Free Newsletter
Sign up for blog updates, science stories, riddles, and occasional musings. No algorithms—just me, writing to you.